Recently, DEW Additive Manufacturing (Taicang) Co., Ltd. (“DEW”) announced that its 17-4PH and 316L stainless steel samples, produced using its in-house Binder Jetting 3D printing technology, have successfully passed in vitro cytotoxicity testing.
The test results indicate that, under the specified test conditions, the samples did not exhibit potential cytotoxicity, meeting the requirements of GB/T 16886.5-2017 and EN ISO 10993-5:2009 for cytotoxicity evaluation.
The tested samples were manufactured using DEW’s proprietary Binder Jetting systems, and the evaluation was conducted by a CNAS-accredited third-party testing laboratory.
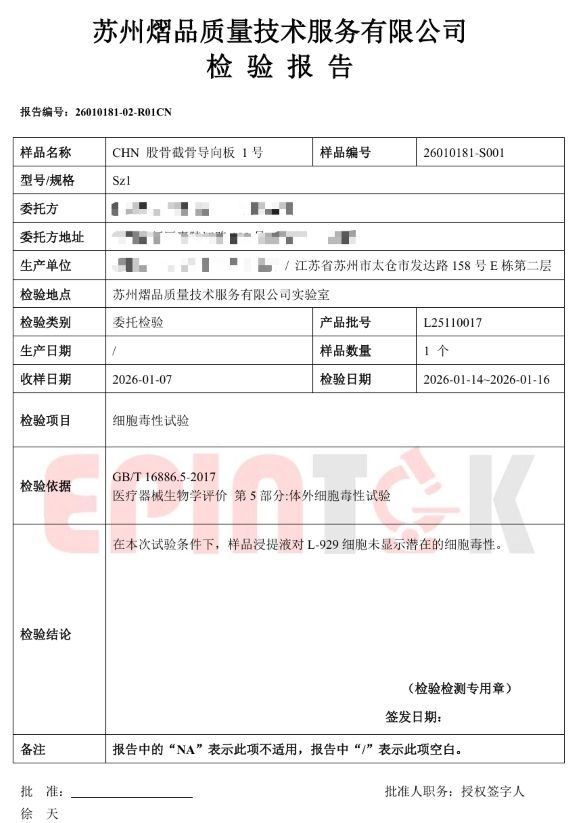
Fig. 1 Cytotoxicity test report for 17-4PH sample
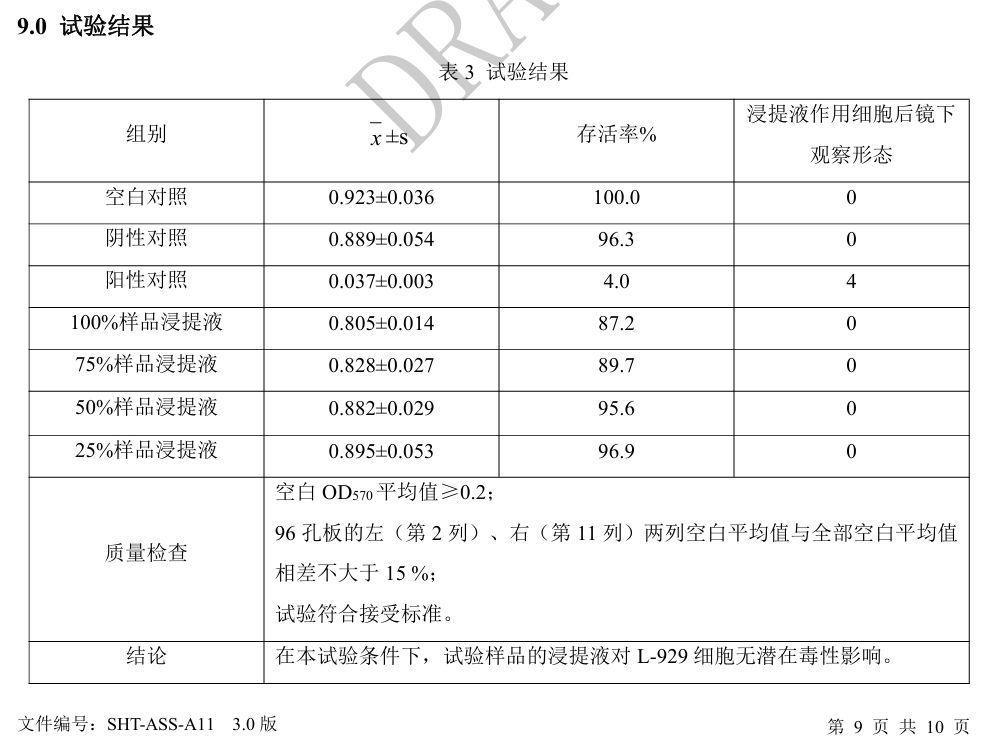
Fig. 2 Cytotoxicity test results for 316L sample
The testing was performed in accordance with YY/T 0290.5-2023 and ISO 10993-5 standards. In vitro cytotoxicity testing is a fundamental assessment within the biological evaluation framework for medical materials, providing an initial indication of potential effects on cell viability and growth.
These results provide baseline data supporting further investigation of Binder Jetting for metal components intended for applications involving contact with the human body.

Fig. 3 DEW Binder Jetting systems: S200 and S400
Binder Jetting, as a powder-bed-based metal additive manufacturing process, offers distinct advantages in producing complex geometries and enabling small-batch and customized production. The process typically does not require support structures, which can reduce post-processing steps and supports a high level of automation.

Fig. 4 Binder Jetting parts produced by DEW
In recent years, DEW has focused on continuous optimization of material systems, equipment stability, and sintering processes, accumulating practical experience in the processing of commonly used stainless steels such as 17-4PH and 316L.
Through iterative improvements in binder formulation, powder characteristics, and sintering pathways, DEW has enhanced both structural integrity and production consistency, supporting further development of applications in demanding industrial sectors.
The completion of in vitro cytotoxicity testing for these stainless steel samples provides supporting data for ongoing research and development of Binder Jetting in medical-related applications, and serves as a foundation for subsequent, more comprehensive biological evaluations.
Looking ahead, DEW will continue advancing Binder Jetting technology across multiple material systems, including:
- Stainless steels
- Copper
- Cemented carbides
- Silicon carbide
The company will further explore applications in medical devices, thermal management, and high-end manufacturing, leveraging the design freedom of additive manufacturing to enable integrated functional and structural components.












